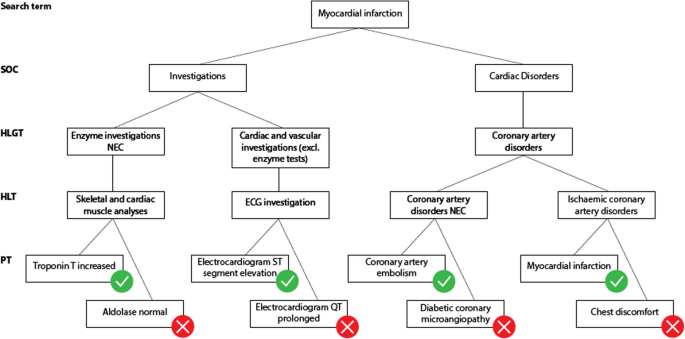
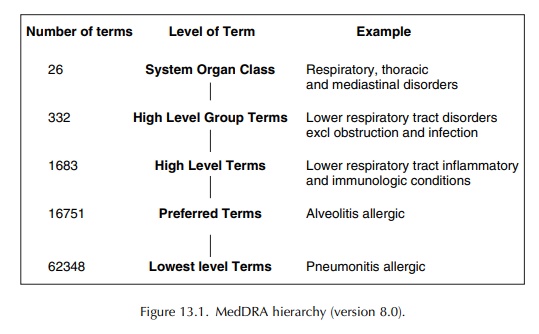
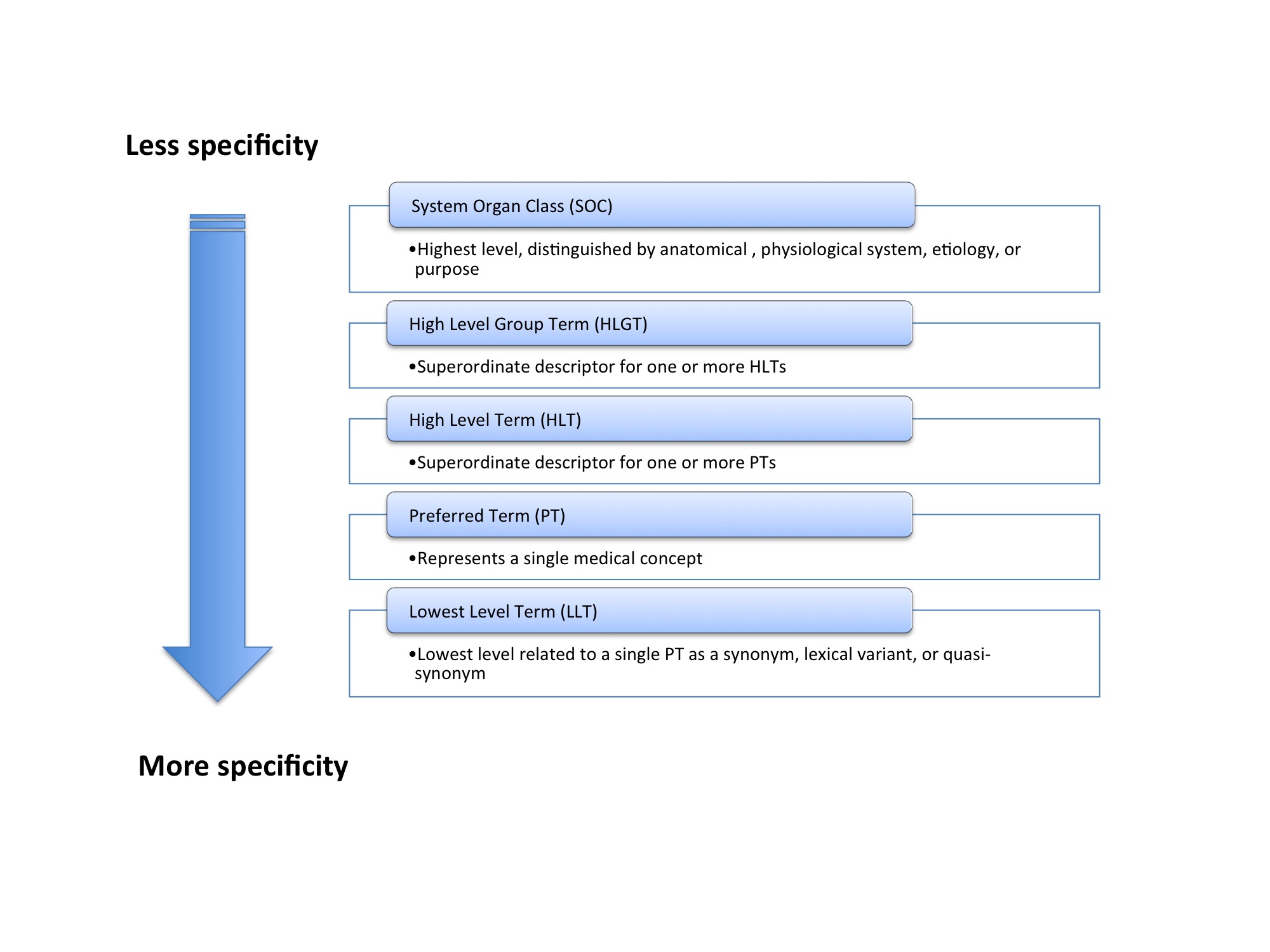
How to interact with medical terminologies? Formative usability evaluations comparing three approaches for supporting the use of MedDRA by pharmacovigilance specialists | BMC Medical Informatics and Decision Making | Full Text
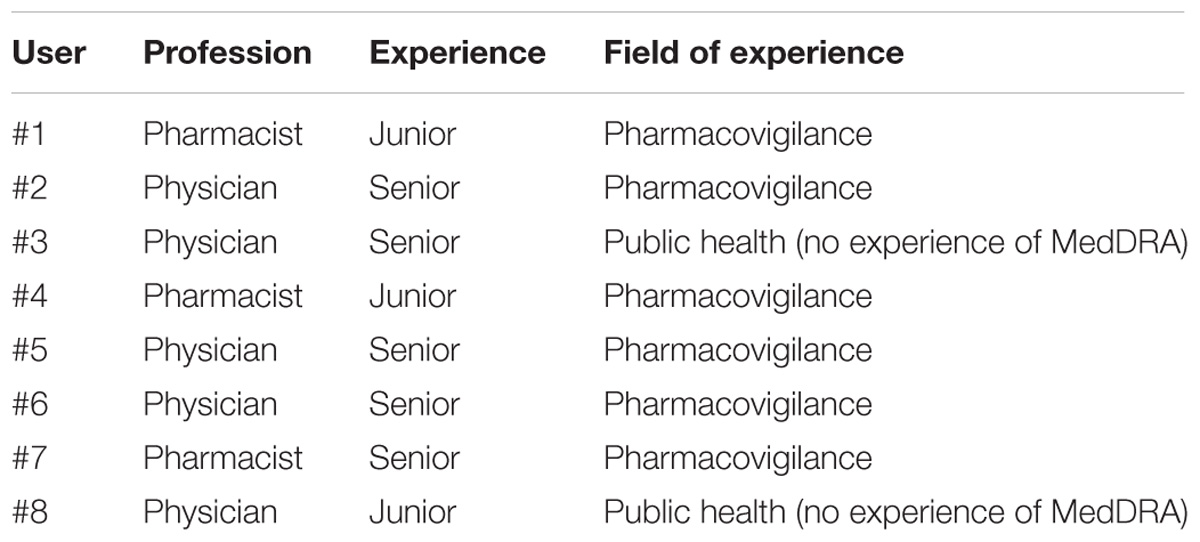
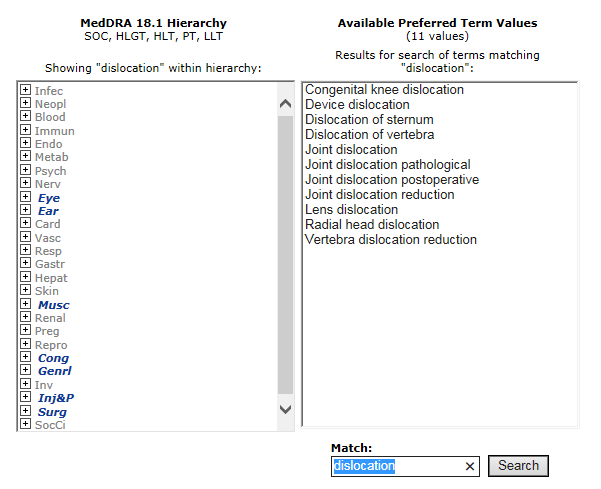
Frontiers | Semantic Queries Expedite MedDRA Terms Selection Thanks to a Dedicated User Interface: A Pilot Study on Five Medical Conditions
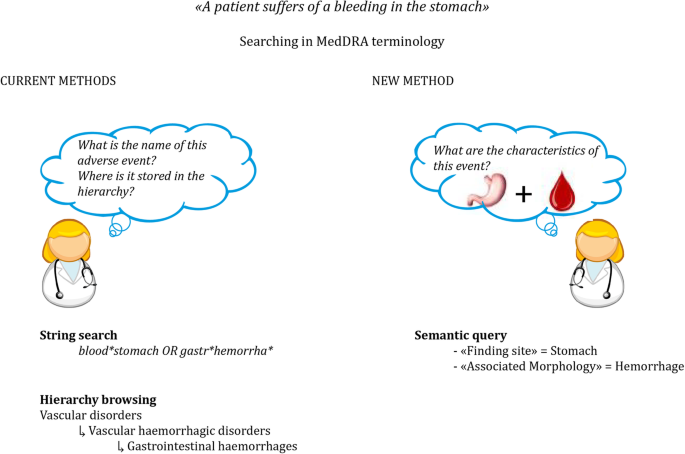




.png)
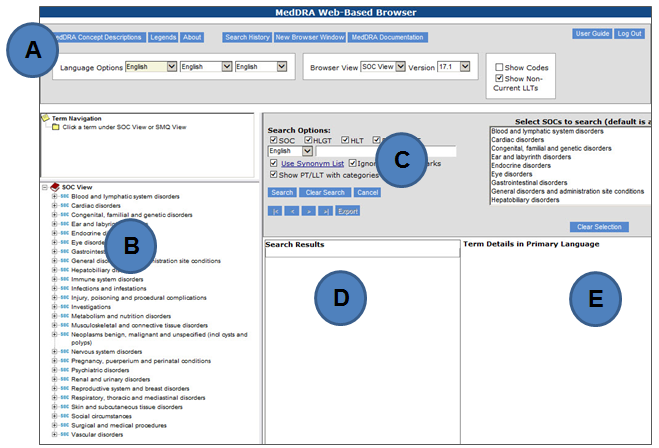

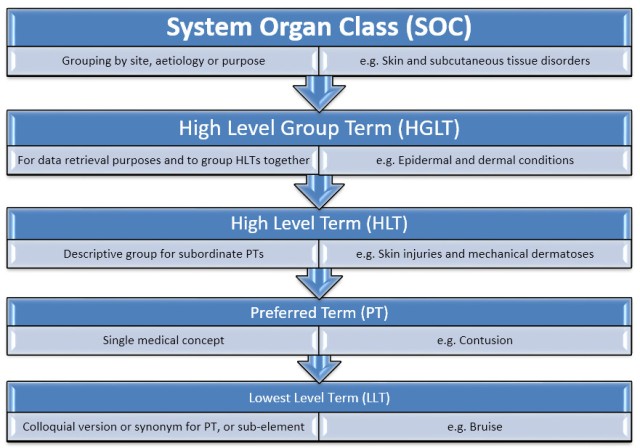
![PDF] Challenges in Coding Adverse Events in Clinical Trials: A Systematic Review | Semantic Scholar PDF] Challenges in Coding Adverse Events in Clinical Trials: A Systematic Review | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/052853043e0670408e8276261478c5b7eb903f11/4-Table1-1.png)





